Select Currency
Bioseparation & Electrophoresis
-
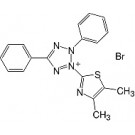
(4,5-Dimethyl-2-thiazolyl)-2,5-diphenyl-2H-tetrazolium-bromide research grade (20395)
Starting at: £18.85

-
-

-

-

-

-

-

-

-

-

-

-

-
-

-
1D SDS NetGel NF 12.5% 25S Kit contains 4 gels (43899) and buffer kit (43383) (43500.01)
£438.46Product Size: 1KIT
-

-

-

-
2D Gel DALTsix 10 -15 % Kit contains 6 gels (43878) and buffer kit (43325) (43318.01)
£509.89Product Size: 1KIT
-
2D Gel DALTsix 12.5% Kit contains 6 gels (43877) and buffer kit (43325) (43317.01)
£509.89Product Size: 1KIT
-
2D Gel DALTsix NF 10 -15 % Kit contains 6 gels (43869) and buffer kit (43325) (43314.01)
£509.89Product Size: 1KIT
-
2D Gel DALTsix NF 12.5 % Kit contains 6 gels (43868) and buffer kit (43325) (43313.01)
£509.89Product Size: 1KIT
-
2D Gel DALTtwelve 10 -15 % Kit contains 12 gels (43878) and buffer kit (43326) (43320.01)
£863.04Product Size: 1KIT
-
2D Gel DALTtwelve NF 10 -15 % Kit contains 12 gels (43869) and buffer kit (43326) (43316.01)
£863.04Product Size: 1KIT
-
2D Gel DALTtwelve NF 12.5% Kit contains 12 gels (43868) and buffer kit (43326) (43315.01)
£863.04Product Size: 1KIT
-
2D Gel DALTwelve 12.5% Kit contains 12 gels (43877) and buffer kit (43326) (43319.01)
£863.04Product Size: 1KIT
-
2D HPE (TM) Large BlotGel NF 10-15 % Kit contains 4 gels and buffer kit (43312) (43435.01)
£523.78Product Size: 1KIT
-
2D HPE (TM) Large BlotGel NF 12.5 % Kit contains 4 gels and buffer kit (43312) (43432.01)
£523.78Product Size: 1KIT
-
2D HPE (TM)Triple BlotGel NF 12.5 % contains 4 gels (43879) and buffer kit (43312) (43429.01)
£466.24Product Size: 1KIT
-
2D HPE(TM) Double BlotGel NF 12.5% Kit contains 4 gels (43898) and buffer kit (43312) (43430.01)
£466.24Product Size: 1KIT
-
2D HPE(TM) Double Gel 10 - 15% Kit contains 4 gels (43863) and buffer kit (43312) (43309.01)
£350.18Product Size: 1KIT
-
2D HPE(TM) Double Gel 12.5% Kit contains 4 gels (43862) and buffer kit (43312) (43308.01)
£350.18Product Size: 1KIT
-
2D HPE(TM) Double Gel NF 10 - 15% Kit contains 4 gels (43856) and buffer kit (43312) (43303.01)
£350.18Product Size: 1KIT
-
2D HPE(TM) Double Gel NF 12.5% Kit contains 4 gels (43855) and buffer kit (43312) (43302.01)
£350.18Product Size: 1KIT
-
2D HPE(TM) Large Gel 10 - 15% Kit contains 4 gels (43866) and buffer kit (43312) (43311.01)
£424.58Product Size: 1KIT
-
2D HPE(TM) Large Gel 12.5% Kit contains 4 gels (43865) and buffer kit (43312) (43310.01)
£424.58Product Size: 1KIT
-
2D HPE(TM) Large Gel NF 10 - 15% Kit contains 4 gels (43860) and buffer kit (43312) (43305.01)
£424.58Product Size: 1KIT
-
2D HPE(TM) Large Gel NF 12.5% Kit contains 4 gels (43857) and buffer kit (43312) (43304.01)
£424.58Product Size: 1KIT
-
2D HPE(TM) Mercator Gel 12.5 % Kit contains 4 gels (43873) and buffer kit (43312) (43410.01)
£363.07Product Size: 1KIT
-
2D HPE(TM) Triple Gel 10 - 15% Kit contains 4 gels (43882) and buffer kit (43312) (43307.01)
£350.18Product Size: 1KIT
-
2D HPE(TM) Triple Gel 12.5% Kit contains 4 gels (43864) and buffer kit (43312) (43306.01)
£350.18Product Size: 1KIT
-
2D HPE(TM) Triple Gel NF 10 - 15% Kit contains 4 gels (43881) and buffer kit (43312) (43301.01)
£350.18Product Size: 1KIT
-
2D HPE(TM) Triple-Gel NF 12.5% Kit contains 4 gels (43859) and buffer kit (43312) (43300.01)
£350.18Product Size: 1KIT
-

-

-

-

-

-